ThermochemistryHard
Question
Reaction involving gold have been of particular intersect to alchemists. Consider the following reactions,
Au(OH)3 + 4 HCl HAuCl4 + 3H2O, áƒH = - 28 kcal
Au(OH)3 + 4 HBr HAuBr4 + 3H2O, áƒH = - 36.8 kcal
In an experiment there was an absorption of 0.44 kcal when one mole of HAuBr4 was mixed with 4 moles of HCl. What is the percentage conversion of HAuBr4 into HAuCl4 ?
Options
A.0.5%
B.0.6%
C.5%
D.50%
Solution
HAuBr4 + 4HCl HAuCl4 + 4HBr
         áƒH = - 28 + 36.8
             = 8.8 kcal
% converssion is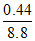 × 100 = 5.
× 100 = 5.
         áƒH = - 28 + 36.8
             = 8.8 kcal
% converssion is
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
The thermochemical equation for the dissociation of hydrogen gas into atoms may be written asH2 → 2H; ΔH = 432.0 kJ.What...When 1.0 g of oxalic acid is burnt in a bomb calorimeter whose heat capacity is 8.75 kJ/K, the temperature increases by ...The enthalpies of formation of SO2(g), H2O(l), HCl(g) and H2SO4(l) are −70.97, −68.32, −22.1 and −188.84 (kcal mol−1). T...The specific heats of iodine vapours and solid are 0.031 and 0.055 cal/g, respectively. If the enthalpy of sublimation o...The polymerization of ethylene to linear polyethylene is represented by the reactionnCH2 = CH2→ (−CH2−CH2−)nwhere n has ...