NEET | 2018Atomic StructureHard
Question
Magnesium reacts with an element (X) to form an ionic compound. If the ground state electronic configuration of (X) is 1s2 2s2 2p3, the simplest formula for this compound is
Options
A.
Mg2X3
B.
MgX2
C.
Mg2X
D.
Mg3X2
Solution
Magnesium ion = Mg+2
X = Nitrogen Nitrogen ion = N–3
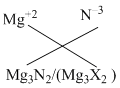
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
What is the angular speed of an electron revolving in the third orbit of He+ ion?...The time period of revolution in the third orbit of Li2+ ion is x second. The time period of revolution in the second or...′e′ configuration of element with minimum size will be :-...The qualitative order of Debroglie wavelength for electron, proton and a particle is λe > λP > λ	...X X1 + H2O → HX1 + HOXSelect the correct statement for above reaction :...