NEET | 2016Aromatic HydrocarbonsHard
Question
Consider the nitration of benzene using mixed conc. H2SO4 and HNO3. If a large amount of KHSO4 is added to the mixture, the rate of nitration will be:
Options
A.
Faster
B.
Slower
C.
Unchanged
D.
Doubled
Solution
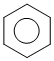 + H2SO4 + HNO3
+ H2SO4 + HNO3 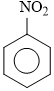 + HSO4- + H2O
+ HSO4- + H2O
If we add KHSO4-, conc. HSO4- increases, equilibrium shifts backward.
Create a free account to view solution
View Solution FreeMore Aromatic Hydrocarbons Questions
Which of the following reactions of benzene does not prove the presence of three carbon − carbon double bonds in i...If p-methoxy toluene is nitrated, the major product is :...Which one of the following compounds undergo bromination of its aromatic ring (electrophilic aromatic substitution) at t...In the sulphonation, acetylation and formylation of benzene the group of effective electrophiles would be:...The major product formed in the reaction + (CH3)2CHCH2Br is−...