JEE Advanced | 2016CalorimetryHard
Question
A water cooler of storage capacity 120 litres can cool water at a constant rate of P watts. In a closed circulation system (as shown schematically in the figure), the water from the cooler is used to cool an external device that generates constantly 3 kW of heat (thermal load). The temperature of water fed into the device cannot exceed 30 0C and the entire stored 120 litres of water is initially cooled to 10 0C. The entire system is thermally insulated. The minimum value of P (in watts) for which the device can be operated for 3 hours is
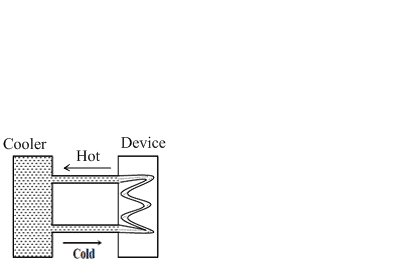
(Specific heat of water is 4.2 kJ kg-1 K-1 and the density of water is 1000 kg m-3 )
Options
A.
1600Â
B.
2067
C.
 2533 Â
D.
3933
More Calorimetry Questions
If I is the moment of inertia of a solid body having a -coefficient of linear expansion then the change in I correspondi...The volume thermal expansion coefficient of an ideal gas at constant pressure is...When m gm of water at 10oC is mixed with m gm of ice at 0oC, which of the following statements are false?...A metal ball immersed in water weighs w1 at 5oC and w2 at 50oC. The coefficient of cubical expansion of metal is less th...An experiment takes 10 minutes to raise the temperature of water in a container from 0oC to 100oCand another 55 minutes ...