CalorimetryHard
Question
An experiment takes 10 minutes to raise the temperature of water in a container from 0oC to 100oC
and another 55 minutes to convert it totally into steam by a heater supplying heat at a uniform rate.
Neglecting the specific heat of the container and taking specific heat of water to be 1 cal / g oC, the
heat of vapourization according to this experiment will come out to be :
and another 55 minutes to convert it totally into steam by a heater supplying heat at a uniform rate.
Neglecting the specific heat of the container and taking specific heat of water to be 1 cal / g oC, the
heat of vapourization according to this experiment will come out to be :
Options
A.560 cal/ g
B.550 cal / g
C.540 cal/ g
D.530 cal/ g
Solution
Pt = mCᐃT
P × 10 × 60 = mC 100
P × 55 × 60 = mL
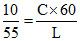
L = 550 cal./g. Option (2)
P × 10 × 60 = mC 100
P × 55 × 60 = mL
L = 550 cal./g. Option (2)
Create a free account to view solution
View Solution FreeMore Calorimetry Questions
Heat given to a body which raises its temperature by 1oC is...A water cooler of storage capacity 120 litres can cool water at a constant rate of P watts. In a closed circulation syst...Two rods having length l1 and l2, made of materials with the linear coefficient of expansion α1 and α2, were w...Two solid spheres A and B of equal volumes but of different densities dA and dB are conected by a string. They are fully...A metal ball immersed in water weighs w1 at 5oC and w2 at 50oC. The coefficient of cubical expansion of metal is less th...