Nitrogen Containing CompoundHard
Question
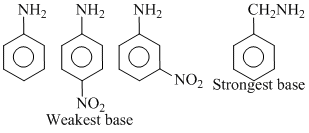
Among the following, the strongest base is :
Options
A.C6H5NH2
B.p-NO2C6H4NH2
C.m-NO2.C6H4NH2
D.C6H5CH2NH2
Solution
In a, b and c, atom is directly attached to benzene ring and its lone pair tart in resonance with nenzene ring and so it is less available for protonation, so they are weaker bases. On the otherhand, in benzylamine lone pair of N-atom can not take part in resonance and so this - NH2 gp. is almost as stronger base as alkyl amino group and so strongest base among these.

Create a free account to view solution
View Solution FreeMore Nitrogen Containing Compound Questions
In the reaction shown below, the major product(s) formed is/are Product(s)...Among the properties (a) reducing (b) oxidising (c) complexing, the set of properties shown by CN- ion towards metal spe...Ammonolysis of R-X gives :-...Propylamine reacts with nitrous acid to from a relatively stable cation viz. -...Examine the following two structures for the anilinium ion and choose the corect statement from the ones given below :...