JEE Main | 2014d and f-Block ElementsHard
Question
Which series of reactions correctly represents chemical reactions related to iron and its compound?
Options
A.Fe FeSO4
FeSO4  Fe2(SO4)3
Fe2(SO4)3 Fe
Fe
 FeSO4
FeSO4  Fe2(SO4)3
Fe2(SO4)3 Fe
FeB.Fe FeO
FeO  FeSO4
FeSO4 Fe
Fe
 FeO
FeO  FeSO4
FeSO4 Fe
FeC.Fe FeCl3
FeCl3  FeCl2
FeCl2 Fe
Fe
 FeCl3
FeCl3  FeCl2
FeCl2 Fe
FeD.Fe Fe3O4
Fe3O4  FeO
FeO Fe
Fe
 Fe3O4
Fe3O4  FeO
FeO Fe
FeSolution
Fe 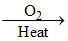 Fe3O4
Fe3O4
This reaction is corresponding to the combustion of Fe.
Fe3O4 FeO
FeO 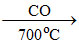 Fe
Fe
These reactions correspond to the production of Fe by reduction of Fe3O4 in blast furnace.
This reaction is corresponding to the combustion of Fe.
Fe3O4
These reactions correspond to the production of Fe by reduction of Fe3O4 in blast furnace.
Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
When copper is heated with conc. HNO3 it produces:...Ferric ion forms a prussian blue coloured ppt. due to :-...Which ore contains both iron and copper :-...Zinc blend structure is obtained when Zn2+ occupies :-...Which one of the following ions exhibits d–d transition and paramagnetism as well ?...