NEET | 2018d and f-Block ElementsHard
Question
Which one of the following ions exhibits d–d transition and paramagnetism as well ?
Options
A.
B.
C.
D.
Solution
Cr+6 diamagnetic
Cr+6 diamagnetic
Mn+7 diamagnetic
Mn+6 paramagnetic 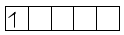
unpaired electron is present so d–d transition is possible.
Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
A red solid is insoluble in water. However it becomes soluble if some KI is added to water. Heating the red solid in a t...The lanthanide contraction is responsible for the fact that...Given below are two statements :Statement I: The number of pairs, from the following, in which both the ions are coloure...When the sample of copper with zinc impurity is to be purified by electrolysis, the appropriate electrodes are...What is wrong about the following pair of compound [Cr(H2O)6]Cl3 and [Cr(H2O)5Cl]Cl2.H2O :-...