Solid StateHard
Question
In an FCC unit cell a cube is formed by joining the centers of all the tetrahedral voids to generate a new cube.Then the new cube would contain voids as :
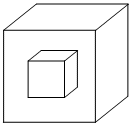

Options
A.1 full Tetrahedral void, 1 full Octahedral void
B.1 full Tetrahedral void only
C.8 full Tetrahedral voids and 1 full Octahedral void
D.1 full Octahedral void only
Solution
No. of tetrahedral voids = 8× 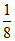 = 1
= 1
No. of Octahedral voids = 1 × 1 = 1 (at body center).
No. of Octahedral voids = 1 × 1 = 1 (at body center).
Create a free account to view solution
View Solution FreeMore Solid State Questions
In the Schoottky defect :...In a face centerd lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers. Then the for...The theoretical density of ZnS is d g/cm3. If the crystal has 4% Frenkel defect, then the actual density of ZnS should b...Which of the following is/are correct ?...Which of the following statement (s) is are correct ?...