SHMHard
Question
For real gases van der Waals equation is written as
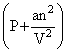 (V - nb) = nRT
(V - nb) = nRT
where ′a′ and ′b′ are van der Waals constants.
I. O2, CO2, H2 and He
II. CH4, O2 and H2
The gases given in set-I in increasing order of ′b′ and gases given in set-II in decreasing order of′a′, are arranged below. Select the correct order from the following:
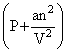 (V - nb) = nRT
(V - nb) = nRTwhere ′a′ and ′b′ are van der Waals constants.
I. O2, CO2, H2 and He
II. CH4, O2 and H2
The gases given in set-I in increasing order of ′b′ and gases given in set-II in decreasing order of′a′, are arranged below. Select the correct order from the following:
Options
A.(I) H2 < O2 < He < CO2 (II) O2 > CH4 > H2
B.(I) He < H2 < CO2 < O2 (II) CH4 > H2 > O2
C.(I) O2 < He < H2 < CO2 (II) H2 > O2 > CH4
D.(I) H2 < He < O2 < CO2 (II) CH4 > O2 > H2
Solution
(I) H2 < He < O2 < CO2 (II) CH4 > O2 > H2
Create a free account to view solution
View Solution FreeMore SHM Questions
A particle free to move along the x-axis has potential energy given by U(x) = k[1 - exp( - x2)] for - ∞ ≤ x ...A particle is describing SHM with amplitude ′a′. When the potential energy of particle is one fourth of the ...The thermal dissociation equilibrium of CaCO3(s) is studied under different conditions.CaCO3(s) ⇋ CaO(s) + CO2(g)F...For a body executing SHM with amplitude A, time period T, maximum velocity vmax and phase constant zero, which of the fo...Which of the following function represents a simple harmonic oscillation :-...