Photoelectric EffectHard
Question
A proton and an electron are accelerated by the same potential difference. Let λe and λp denote the de Broglie wavelengths of the electron and the proton respectively.
Options
A.λe = λp
B.λe < λp
C.λe > λp
D.λe and λp depends on the accelerating potential difference.
Solution
Charge present on both panticle are same
∵ λ =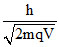 So q1 = q2
So q1 = q2
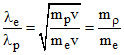 > 1
> 1
∵ mP > > me
∴ λe > λP
∵ λ =
∵ mP > > me
∴ λe > λP
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
The ionisation energies of K-shell for cobalt, copper, and molebdenum are 7.8, 9.0 and 20.1 keV respectively. If any met...If a0 is the Bohr radius, the radius of the n = 2 electronic orbit in triply ionized beryllium is -...Light of two different frequencies whose photons have energies 1 eV and 2.5 eV respectively illuminate a metallic surfac...A photon of light enters a block of glass after travelling through vacuum. The energy of the photon on entering the glas...The energy difference between the first two levels of hydrogen atom is 10.2 eV. What is the corresponding energy differe...