Photoelectric EffectHard
Question
The energy difference between the first two levels of hydrogen atom is 10.2 eV. What is the corresponding energy difference for a singly ionized helium atom ?
Options
A.10.2 eV
B.20.4 eV
C.40.8 eV
D.81.6 eV
Solution
En = 13.6 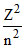
ᐃ EH =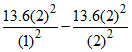 = 10.2eV
= 10.2eV
ᐃEHe =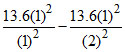 = 40.8 eV
= 40.8 eV
ᐃ EH =
ᐃEHe =
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
The energy of characteristic X-rays photon obtained from coolidge tube comes from :-...In an electron gun electron are accelerated through a potential difference V. if e = charge of electron and m = mass of ...In an α-decay the Kinetic energy of a particle is 48 MeV and Q-value of the reaction is 50 MeV. The mass number of ...If the mass of neutron = 1.7 × 10-27 kg. then the De broglie wavelength of neutron of energy 3eV is :...Two lamps of luminous intensity of 8 Cd and 32 Cd respectively are lying at a distance of 1.2m from each other. Where sh...