Photoelectric EffectHard
Question
In an x-ray tube the voltage applied is 20KV. The energy required to remove an electron from L shell is 19.9 KeV. In the x-rays emitted by the tube (hc = 12420 ev(Ao))
Options
A.minimum wavelength will be 62.1 pm
B.energy of the characterstic x-rays will be equal to or less than 19.9 KeV
C.Lαx-ray may be emitted
D.Lα x-ray will have energy 19.9 KeV
Solution
(A) Minimum wavelength will correspond to maximum energy i.e.,from ∞ to k.
ᐃE = 19.9 KeV
∴ λmin =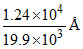 = 0.62 (Ao)
= 0.62 (Ao)
= 62 pm.
(B) Energy of the characteristic x-rays will be less than corresponding to ∞ to k-shell, hence than 19.9 KeV.
ᐃE = 19.9 KeV
∴ λmin =
= 62 pm.
(B) Energy of the characteristic x-rays will be less than corresponding to ∞ to k-shell, hence than 19.9 KeV.
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
The ratio of the wavelengths of the longest wavelength lines in the Lyman and Balmer series of hydrogen spectrum is :...A sphere of mass M and radius R is falling viscous fluid. The terminal velocity attained by the falling object will be p...A B Ct = 0 N0 0 0 t N1 N2 N3In the above radioactive decay C is stable nucleus. Then:...Which experiment explains the wave nature of electron :-...Two separate monochromatic light beams A and B of the same intensity are falling normally on a unit area of a metallic s...