Photoelectric EffectHard
Question
A 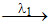 B
B 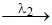 C
C
t = 0 N0 0 0
t N1 N2 N3
In the above radioactive decay C is stable nucleus. Then:
t = 0 N0 0 0
t N1 N2 N3
In the above radioactive decay C is stable nucleus. Then:
Options
A.rate of decay of A will first increase and then decrease
B.number of nuclei of B will first increase and then decrease
C.if λ2 > λ1, then activity of B will always be higher than activity of A
D.if λ2 > λ1, then number of nucleus of C will always be less than number of nucleus of B.
Solution
Rate of decay of A keeps on decreasing continuously because concentration of A decreases with time. ⇒ A is false
Initial rate of production of B is λ1N0 and rate of decay is zero. With time, as the number of B atom increase, the rate of its production decrease and its rate of decay increases. Thus the number of nuclei of B will first increase and then decrease. ⇒ B is the correct choice
The initial activity of B is zero whereas initial activity of A is λ1N0 ⇒ C is false.
As time t → ∞ : NA = 0, NB = 0 and NC = N0. ⇒ D is false
Initial rate of production of B is λ1N0 and rate of decay is zero. With time, as the number of B atom increase, the rate of its production decrease and its rate of decay increases. Thus the number of nuclei of B will first increase and then decrease. ⇒ B is the correct choice
The initial activity of B is zero whereas initial activity of A is λ1N0 ⇒ C is false.
As time t → ∞ : NA = 0, NB = 0 and NC = N0. ⇒ D is false
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
A monochromatic source of 25 watt emit photons of wave length 6000then number of photons emitted per second are:-...Bernulli equation is a consequence of conservation of :...92U235 nucleus absorbs a slow neutron and undergoes fission into 54X139 and 38Sr94 nuclei. The other particles produced ...Work of 3.0 × 10-4 joule is required to be done in increasing the size of a soap film from 10cm × 6cm to 10cm ...When the wavelength of incident light is 4000, stopping potential is 2 volt. If the wavelength of light is 3000 then sto...