Redox and Equivalent ConceptHard
Question
In which of the folowing compounds, nitrogen exhibits highest oxidation state ?
Options
A.N2H4
B.NH3
C.N3H
D.NH2OH
Solution
Let the oxidation state of nitrogen in the given compounds be x.]
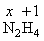
2( x ) + ( +1 )4 = 0
2x = - 4
∴ x = - 2
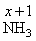
x + ( +1 )3 = 0
∴ x = - 3
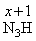
( x )3 + ( +1 ) = 0
3x = - 1
∴ x = - 1/3
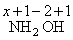
x + ( + 1 )2 + ( - 2 ) + ( + 1 ) = 0
x + 2 - 2 + 1 = 0
x + 1 = 0
∴ x = - 1
Thus, oxidation state of nitrogen is highest in N3H.
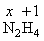
2( x ) + ( +1 )4 = 0
2x = - 4
∴ x = - 2
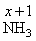
x + ( +1 )3 = 0
∴ x = - 3
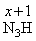
( x )3 + ( +1 ) = 0
3x = - 1
∴ x = - 1/3
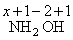
x + ( + 1 )2 + ( - 2 ) + ( + 1 ) = 0
x + 2 - 2 + 1 = 0
x + 1 = 0
∴ x = - 1
Thus, oxidation state of nitrogen is highest in N3H.
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
Which of the following have been arranged in the order of decreasing oxidation number of sulphur?...Which of the following is/are peroxide(s)?...A quantity of 15.8 g of KMnO4 can be decolourized in acidic medium by (K = 39, Mn = 55, Fe = 56)...Expermentally it was found that a metal oxide has formula M0.98O. Metal M, is present as M2+and M3+ in its oxide. Fracti...The number of electrons involved in the reduction of nitrate ion to hydrazine is...