Photoelectric EffectHard
Question
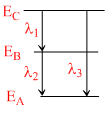
Energy levels A, B and C of a certain atom correspond to increasing values of energy, i.e. EA < EB < EC. If λ1, λ2 and λ3 are the wavelengths of radiations corresponding to transitions C to B, B to A and C to A respectively, which of the following relations is correct ?
Options
A.λ3 = λ1 + λ2
B.λ3 = 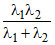
C.λ1 + λ2 + λ3 = 0
D.λ32 = λ12 + λ22
Solution
EC − EA = (EC − EA) + (EB − EA)
⇒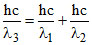
⇒ λ3 =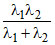
⇒
⇒ λ3 =

Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
What is the momentum of a photon having frequency 1.5 × 1013 Hz :...Which of the following is a wrong description of binding energy of a nucleus ?...Chemical to electricalWhich one of the following is true in photoelectric emission :-...X-ray is an electromagnetic radiation, so X-ray photons carry :...Two small drops of mercury, each of radius R coalesece to form a single large energies before and after the change is :...