Photoelectric EffectHard
Question
If an orbital electron of the hydrogen atom jumps from the ground state to a higher energy state, its orbital speed reduces to half its initial value. If the radius of the electron orbit in the ground state is r, then the radius of the new orbit would be :
Options
A.2r
B.4r
C.8r
D.16r
Solution
Since speed reduces to half , KE reduced to
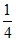 th ⇒ n = 2
th ⇒ n = 2
mvr =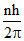
mv0 r = 1.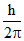 ..........I
..........I
m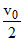 r′= 2
r′= 2 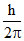 ..........II
..........II
r′ = 4.r
mvr =
mv0 r = 1.
m
r′ = 4.r
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
When a β−-particle is emitted from a nucleus, the neutron-proton ratio :...The energy of characteristic X-rays photon obtained from coolidge tube comes from :-...Energy levels A, B and C of a certain atom correspond to increasing values of energy, i.e. EA < EB < EC. If λ...For producing the X-rays, what is the value of voltage (nearly), which is applied on the tube:...Candela is the unit of :...