KTGHard
Question
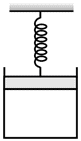
One mole of an ideal gas is kept enclosed under a light piston (area=10-2 m2) connected by a compressed spring (spring constant 100 N/m). The volume of gas is 0.83 m3 and its temperature is 100K. The gas is heated so that it compresses the spring further by 0.1 m. The work done by the gas in the process is: (Take R = 8.3 J/K-mole and suppose there is no atmosphere).

Options
A.3 J
B.6 J
C.9 J
D.1.5 J
Solution
Before heating let the pressure of an ideal gas be P1 from the equilibrium piston,
P1A = kx1 [For equilibrium position]
∴ x1 =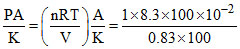
= 0.1 m
Since during heating process,
The spring is compressed further by 0.1 m
∴ x2 = 0.2 m
work done by gas =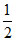 .100(0.22 − 0.12) =
.100(0.22 − 0.12) = 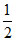 .100.(0.1) (0.3)
.100.(0.1) (0.3)
= 1.50 = 1.5 J
P1A = kx1 [For equilibrium position]
∴ x1 =
= 0.1 m
Since during heating process,
The spring is compressed further by 0.1 m
∴ x2 = 0.2 m
work done by gas =
= 1.50 = 1.5 J
Create a free account to view solution
View Solution FreeMore KTG Questions
One mole of an ideal gas at a temperature T1K expands slowly according to the law = constant. Its final temperature is T...What is the meaning of point masses for the molecules of ideal gas :-...Relation between pressure (P) and energy density (E) of an ideal gas is -...The temperature of H2 and N2 are respectively 300 K and 450 K. The ratio of their average kinetic energies will be:-...When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the ...