KTGHard
Question
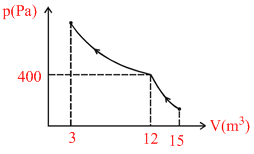
Curve in the figure shows an adiabatic compression of an ideal gas from 15 m3 to 12 m3, followed by an isothermal compression to a final volume of 3.0 m3. There are 2.0 moles of the gas. Total heat supplied to the gas is equal to : (ln2 = 0.693)

Options
A.4521 J
B.− 4521 J
C.− 6654 J
D.− 8476 J
Solution
There is no heat transfer in adiabatic compression. In isothermal process
Q = W = P1V1 ln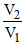
= 400 × 12 ln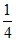 = − 6654 J
= − 6654 J
Q = W = P1V1 ln
= 400 × 12 ln
Create a free account to view solution
View Solution FreeMore KTG Questions
A diatomic ideal gas undergoes a thermodynamic change according to the P−V diagram shown in the figure. The total ...The total kinetic energy of 1 mole of N2 at 27o C will be approximately :-...The variation of PV graph with V of a fixed mass of a ideal gas at constant temperature is graphically represented as sh...Temperature at which the velocity of sound in O2 is the same as that on N2 at 27oC is approximately...In given figure a cylindrical vessel contains nitrogen and closed by a 20 kg frictionless piston. If cylinder is slowly ...