KTGHard
Question
A mass of an ideal gas undergoes a reversible isothermal compression. Its molecules will then have compared with initial state, the same
(i) root mean square velocity (ii) mean mometum (iii) mean kinetic energy
(i) root mean square velocity (ii) mean mometum (iii) mean kinetic energy
Options
A.(i), (ii), (iii) correct
B.(i), (ii) correct
C.(ii), (iii) correct
D.(i) correct
Solution
As initial and final state are same
∴ TI = TF As Vrms,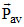 and
and 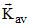
depends on temperature
∴ all are equal.
∴ TI = TF As Vrms,
depends on temperature
∴ all are equal.
Create a free account to view solution
View Solution FreeMore KTG Questions
N2 molecules is 14 times heavier than a H2 molecule. At what temperature will the rms speed of H2 molecules be equal to ...In a cyclic process shown on the P − V diagram the magnitude of the work done is :...At absolute zero temperature :-...If temperature is changed from 27oC to 327oC. The ratio of K.E. molecules at two temperature....When an ideal gas undergoes an adiabatic change causing a temperature change ᐃT(i) there is no heat gained or lost...