KTGHard
Question
The quantity 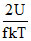 represents (where U = internal energy of gas)
represents (where U = internal energy of gas)
Options
A.mass of the gas
B.kinetic energy of the gas
C.number of moles of the gas
D.number of molecules in the gas
More KTG Questions
The temperature of the hydrogen at which the average speed of its molecules is equal to that of oxygen molecules at a te...The speeds of 5 molecules of a gas (in arbitary units) are as follows 2,3,4,5,6 The root mean square speed for these moe...Equal volume of H2, O2 and He gases are at same temperature and pressure. Which of these will have large number of molec...The given curve represents the variation of temperature as a function of volume for one mole of an ideal gas. Which of t...A thermodynamic process of one mole ideal mono atomic gas is shown in figure. The efficiency of cyclic process ABCA will...