KTGHard
Question
The molar heat capacity for the process shown in fig. is
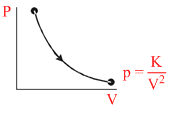

Options
A.C = CV
B.C = CP
C.C > CV
D.C < CV
Solution
For polytropic process PVx = k;
C = CV +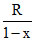 ⇒ As PV2 = K (given) ⇒ Put x = 2
⇒ As PV2 = K (given) ⇒ Put x = 2
C = CV +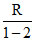 = CV − R. ∴ C < CV.
= CV − R. ∴ C < CV.
C = CV +
C = CV +
Create a free account to view solution
View Solution FreeMore KTG Questions
The specific heat of an ideal gas depends on temperature is-...Hydrogen and helium volume V at same temperature T and same pressure P are mixed to have same volume V. The resulting pr...One mole of an ideal gas undergoes a process in which T = T0 + aV3, where T0 and ′a′ are positive constants ...Which of the follwing will be same if temperature of two ideal gas is equal...Find the ratio of specific heat at constant pressure to the specific heat at constant volume for NH3 :-...