KTGHard
Question
In figure, an ideal gas is expanded from volume V0 to 2V0 under three different processes. Process 1 is isobaric, process 2 is isothermal and process 3 is adiabatic. Let ᐃU1,ᐃU2 and ᐃU3 be the change in internal energy of the gas is
these three processes. Then :
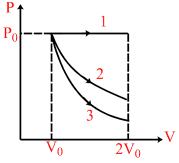
these three processes. Then :

Options
A.ᐃU1 > ᐃU2 > ᐃU3
B.ᐃU1 < ᐃU2 < ᐃU3
C.ᐃU2 < ᐃU1 < ᐃU3
D.ᐃU2 < ᐃU3 < ᐃU1
Solution
Process ...(1) is isobaric
ᐃU1 = ᐃQ − ᐃW = positive
process (2) is isothermal
ᐃU2 = 0
Process (3) is adiabatic
ᐃQ = 0
ᐃU = − ᐃW = negative
∴ ᐃU1 > ᐃU2 > ᐃU3
ᐃU1 = ᐃQ − ᐃW = positive
process (2) is isothermal
ᐃU2 = 0
Process (3) is adiabatic
ᐃQ = 0
ᐃU = − ᐃW = negative
∴ ᐃU1 > ᐃU2 > ᐃU3
Create a free account to view solution
View Solution FreeMore KTG Questions
A one dimensional gas is a hypothetical gas with molecules that can move along only a single axis. The following table g...N molecules of an ideal gas at temperature T1 and presseure P1 are contained in a closed box. If the molecules in the bo...The r.m.s speed of oxygen molecules at $47^{\circ}C$ is equal to that of the hydrogen molecules kept at $\_\_\_\_$ $\ ^{...The wrong statement according to the kinetic theory of gases is -...N2 molecules is 14 times heavier than a H2 molecule. At what temperature will the rms speed of H2 molecules be equal to ...