KTGHard
Question
When an enclosed perfect gas is subjected to an adiabatic process:
Options
A.When an enclosed perfect gas is subjected to an adiabatic process:
B.Its temperature does not change
C.Its pressure varies inversely as a certain power of its volume
D.The product of its pressure and volume is directly proportional to its absolute temperature.
Solution
For adiabatic process PVγ = K.
or P μ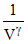
And PV = nRT
or P μ
And PV = nRT
Create a free account to view solution
View Solution FreeMore KTG Questions
Boiling water is changing into steam. Under this condition, the specific heat of water is...The molar heat capacity for the process shown in fig. is...The internal energy of an ideal gas depends on -...The lowest pressure (the best vaccum) that can be created in laboratory at 27oC is 10-11 mm of Hg. At this pressure, the...For an ideal gas molecules the following quantities having zero average values are....