KTGHard
Question
When a system is taken from state ′a′ to state ′b′ along the path ′acb′, it is found that a quantity of heat Q = 200 J is absorbed by the system and a work W = 80J is done by it. Along the path ′adb′, Q = 144J. The work done along the path ′adb′ is
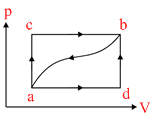

Options
A.6J
B.12J
C.18J
D.24J
Solution
ᐃU = same is both process
Qacb − Wacb = Qadb − Wadb .
200 − 80 = 144 − Wadb. Wadb = 24 J.
Qacb − Wacb = Qadb − Wadb .
200 − 80 = 144 − Wadb. Wadb = 24 J.
Create a free account to view solution
View Solution FreeMore KTG Questions
If one mole of a mono-atomic gas (γ = 5/3) is mixed with one mole of a diatomic gas (γ = 7/5), the value of g ...In an isothermal expansion of an ideal gas. Select wrong statement:...Let P1 and P2 be the final pressure of the samples 1 and 2 respectively in the previous question then :...The speed of sound in oxygen (O2) at a certain temperature is 460 ms-1. The speed of sound in helium (He) at the same te...Specific heat of a substance can be :-...