KTGHard
Question
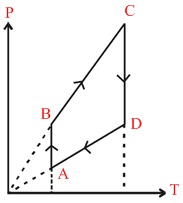
Pressure versus temperature graph of an ideal gas is as shown in figure.

Options
A.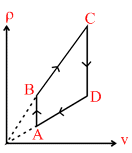

B.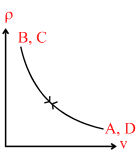

C.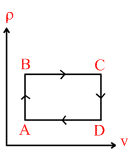

D.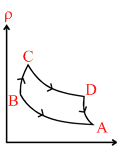

Solution
As PV = nRT m = ρV = constant or ρ α 1/V
PV = nRT m = ρV = P α ρ
A → B T = constant, pressure increases or volume decreases
B → C Volume is constant, V = constant
C → D P is decreases or volume increases [T = constant]
D → A Volume is constant V = with constant,
clearly option ′B′ is constant.
PV = nRT m = ρV = P α ρ
A → B T = constant, pressure increases or volume decreases
B → C Volume is constant, V = constant
C → D P is decreases or volume increases [T = constant]
D → A Volume is constant V = with constant,
clearly option ′B′ is constant.
Create a free account to view solution
View Solution FreeMore KTG Questions
At which temperature of the following, does any gas has average moleculer kinetic energy double that of at 20o C...A gas undergoes a process in which its pressure P and volume V are related as VPn = constant. The bulk modulus of the ga...For an ideal gas molecules the following quantities having zero average values are....According to kinetic theory of gases, at a given temperature:-...An ideal gas at 1 atmospheric pressure and at 273 K has 22.4 litre of volume. This is heated to 546 K and then by applyi...