KTGHard
Question
A gas undergoes a process in which its pressure P and volume V are related as VPn = constant. The bulk modulus of the gas in the process is :
Options
A.nP
B.P1/n
C.P/n
D.Pn
Solution
(C)
VPn = constant.
dV Pn + VnPn-1 dP = 0
−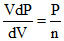 = bulk modulus
= bulk modulus
VPn = constant.
dV Pn + VnPn-1 dP = 0
−
Create a free account to view solution
View Solution FreeMore KTG Questions
Which of the follwing will be same if temperature of two ideal gas is equal...For one gram mole of a gas, the gas equation is PV = RT, here ′R′ is :-...For free expansion of a gas in an adiabatic container which of the following is true ?...Which of the following statement is true according to kinetic theory of gases?...At 0o C temperature root mean square speed of which of the following gases will be maximum:-...