Permutation and CombinationHard
Question
Molar conductivities 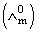 at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively.
at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. 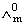 for CH3COOH will be:
for CH3COOH will be:
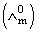 at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively.
at infinite dilution of NaCl, HCl and CH3COONa are 126.4, 425.9 and 91.0 S cm2 mol-1 respectively. 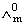 for CH3COOH will be:
for CH3COOH will be:Options
A.390.5 S cm2 mol-1
B.425.5 S cm2 mol-1
C.180.5 S cm2 mol-1
D.290.8 S cm2 mol-1
More Permutation and Combination Questions
How many ways are there to arrange the letters in the word GARDEN with the vowels in alphabetical order?...The number of permutations that can be formed by arranging all the letters of the word ′NINETEEN′ in which n...In how many ways a team of 11 be chosen from 20 students of a class so that 2 particular students are always included an...How many words can be formed by taking three letters from the letters of the word ′SERIES′ ?...The number of words which can be formed from the letters of the word ′SCHOLAR′ which begin with A and end wi...
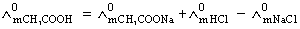 = 91.0 + 425.9 - 126.4 = 390.5
= 91.0 + 425.9 - 126.4 = 390.5