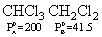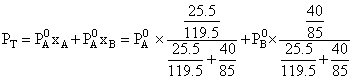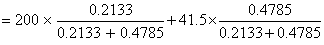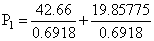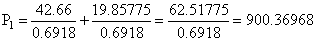DeterminantHard
Question
Vapour pressure of chloroform (CHCl3) and dichloromethane (CH2Cl2) at 25oC are 200 mm Hg and 41.5 mm Hg respectively. Vapour pressure of the solution obtained by mixing 25.5 g of CHCl3 and 40 g of CH2Cl2 at the same temperature will be:
(Molecular mass of CHCl3 = 119.5 u and molecular mass of CH2Cl2 = 85 u)
(Molecular mass of CHCl3 = 119.5 u and molecular mass of CH2Cl2 = 85 u)
Options
A.285.5 mm Hg
B.173.9 mm Hg
C.615.0 mm Hg
D.347.9 mm Hg
More Determinant Questions
If D(x) = , then the value of |ln cos (ᐃx)| will be -...If the system of equation x - ky - z = 0, kx - y - z = o, x + y - z = 0 has a non-zero solution, then possible values of...If a, b, and c, nonzero real numbers, then is equal to -...The number of different possible orders of matrices having 12 identical elements is...If ax + by + cz = 1, bx + cy + az = 0 = cx + ay + bz, then is equal to -...