CalorimetryHard
Question
A metal ball of specific gravity 4.5 and specific heat 0.1 cal/gm-oC is placed on a large slab of ice at 0oC. Half of the ball sinks in the ice. The initial temperature of the ball is :-
(Latent heat capacity of ice = 80 cal/g, specific gravity of ice = 0.9)
(Latent heat capacity of ice = 80 cal/g, specific gravity of ice = 0.9)
Options
A.100oC
B.90oC
C.80oC
D.70oC
Solution
V × 4.5 × 103 × 0.1 × T = 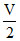 × 0.9 × 103 × 80
× 0.9 × 103 × 80
T =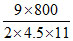 = 80ºC.
= 80ºC.
T =
Create a free account to view solution
View Solution FreeMore Calorimetry Questions
Steam at 100o C is passed into 1.1 kg of water contained in a calorimeter of water equivalent 0.02 kg at 15o C till the ...A small quantity, mass m, of water at a temperature q (inoC) is poured on to a large mass M of ice which is at its melti...When two non reactive samples at different temperatures are mixed in an isolated container of negligible heat capacity t...If a bimetallic strip is heated, it will...If I is the moment of inertia of a solid body having a -coefficient of linear expansion then the change in I correspondi...