CalorimetryHard
Question
A small quantity, mass m, of water at a temperature q (inoC) is poured on to a large mass M of ice which is at its melting point. If c is the specific heat capacity of water and L the latent heat of fusion of ice, then the mass of ice melted is given by :
Options
A.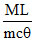
B.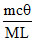
C.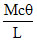
D.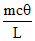
More Calorimetry Questions
A liquid with coefficient of volume expansion γ is filled in a container of a material having the coefficient of li...Two rods having length l1 and l2, made of materials with the linear coefficient of expansion α1 and α2, were w...When m gm of water at 10oC is mixed with m gm of ice at 0oC, which of the following statements are false?...In Newtonᐃs law of cooling, = -k (θ - θ), the constant k is proportional to...Two solid spheres A and B of equal volumes but of different densities dA and dB are conected by a string. They are fully...