Hydrogen and its compoundHard
Question
Fe3+ is reduced to Fe2+ by using -
Options
A.H2O2 in presence of NaOH
B.Na2O2 in water
C.H2O2 in presence of H2SO4
D.Na2O2 in presence of H2SO4
Solution
In acidic medium H2O2 oxidises Fe+2 to Fe+3
In alkaline solution H2O2 reduces Fe+3 to Fe+2
case-1
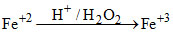
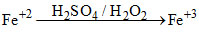
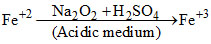
case-2
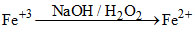
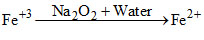
{Na2O2 + 2H2O → 2NOH + H2O2}
In alkaline solution H2O2 reduces Fe+3 to Fe+2
case-1
case-2
{Na2O2 + 2H2O → 2NOH + H2O2}
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
The volume of water which must be added to a mixture of 350 cm3 of 6M HCl and 650 ml of 3M HCl to get a resulting soluti...Concentrated hydrochloric acid when kept in open air sometimes produces a cloud of white fumes. The explanation for it i...Anhydrous ferric chloride is preapred by :...The product obtained by treating benzene with chlorine in presence of ultraviolet light is :...The natural gas mainly contains :...