Work, Power and EnergyHard
Question
A container of fixed volume has a mixutre of one mole of hydrogen and one mole of helium in equilibrium at temperature T. Assuming the gases are ideal, the correct statement(s) is (are) :-
Options
A.The average energy per mole of the gas mixture is 2RT.
B.The ratio of speed of sound in the gas mixture to that in helium gas is 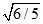
C.The ratio of the rms speed of helium atoms to that of hydrogen molecules is 1/2.
D.The ratio of the rms speed of helium atoms to that of hydrogen molecules is 1/√2.
Solution
CP(mix) = 3R γmix = 3/2 ⇒ f = 4
Average energy/mole = f 1/2 RT = 2RT
∴ (A,B,D)
Create a free account to view solution
View Solution FreeMore Work, Power and Energy Questions
When a conservative force does positive work on a body, then...A particle of mass m is moving in a circular path of constant radius r such that its centrip-etal acceleration ac is var...A capacitor of 2μF is charged as shown in the diagram. When the switch S is turned to position 2, the percentage of...A collision is said to be perfectly inelastic when :-...Two identical balls, one moves with 12 m/s and second is at rest, collides elastically. After collision velocity of seco...