Chemical BondingHard
Question
The geometry of XeOF4 by by VSEPR theory is :
Options
A.pentagonal planar
B.octahedral
C.square pyramidal
D.trigonal bipyramidal
Solution
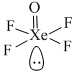 sp3d2 hybridized with one position occupied by lone pair.
sp3d2 hybridized with one position occupied by lone pair.Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Four diatomic species are listed below. Identify the correct order in which the bond order is increasing:...Which of the following has the lowest diploe moment ?...Match the compounds given in column I with the hybridization and shape given in column II and mark the correct option....Which element has high electron affinity ?...Which of the following structure is similar to graphite?...