AIPMT | 2015Nitrogen Containing CompoundHard
Question
In Duma′s method for estimation of nitrogen, 0.25 g of an organic compound gave 40 mL of nitrogen collected at 300 K temperature and 725 mm pressure. If the aqueous tension at 300 K is 25 mm, the percentage of nitrogen in the compound is :-
Options
A.18.20
B.16.76
C.15.76
D.17.36
Solution
Volume of nitrogen collected at 300 K and 725 mm pressure is 40 mL actual pressure
= 725 - 25 = 700 mm
Volume of nitrogen at STP
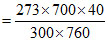 = 33.52mL
= 33.52mL
22,400 mL of N2 at STP weight = 28 g
33.5 mL of nitrogen weight =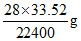
Percentage of nitrogen
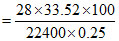 = 16.76%
= 16.76%
= 725 - 25 = 700 mm
Volume of nitrogen at STP
22,400 mL of N2 at STP weight = 28 g
33.5 mL of nitrogen weight =
Percentage of nitrogen
Create a free account to view solution
View Solution FreeMore Nitrogen Containing Compound Questions
The product formed in the reaction...Nitro benzene on treatment with zinc ust and aqueous ammonium chloric gives:...Diethyl oxalate is used for distinguishing primary, secondary and tertiary...The compound obtained by the reaction between primary amine and aldehyde is-...Primary amines on being heated with CS2 in the presence of HgCl2 form alkyl/aryl isothiocyanates. The reaction is known ...