Nitrogen Containing CompoundHard
Question
Diethyl oxalate is used for distinguishing primary, secondary and tertiary
Options
A.alcohols
B.amines
C.alkyd halides
D.hydrogens in hydrocarbons
Solution
Deithyl oxalate is used for distinguidhing 1o, 2o and 3o amines as three amines reacts differently as discussed below.
The 1o amineforms corresponnding subxtituited oxamide which is a crystalline solid while 2o amine forms a diehyl oxamic eater which is a liquid and 3o amine does not react with diethyl oxalate since it does not contain a replacable hyrogen atom.
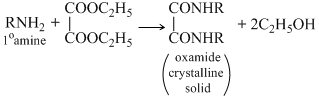
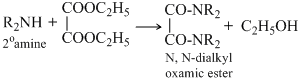
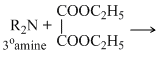 No reaction
No reaction
The 1o amineforms corresponnding subxtituited oxamide which is a crystalline solid while 2o amine forms a diehyl oxamic eater which is a liquid and 3o amine does not react with diethyl oxalate since it does not contain a replacable hyrogen atom.
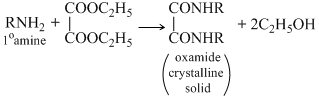
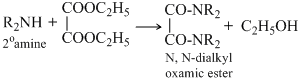
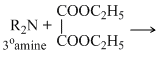 No reaction
No reactionCreate a free account to view solution
View Solution Free