Aromatic HydrocarbonsHard
Question
Consider the following cell reaction:
2Fe(s) + O2(g) + 4H+(aq) → 2Fe2+(aq) + 2H2O(l) Eo = 1.67V
At [Fe2+] = 10-3 M, P(O2) = 0.1 atm and pH = 3, the cell potential at 25oC is
2Fe(s) + O2(g) + 4H+(aq) → 2Fe2+(aq) + 2H2O(l) Eo = 1.67V
At [Fe2+] = 10-3 M, P(O2) = 0.1 atm and pH = 3, the cell potential at 25oC is
Options
A.1.47 V
B.1.77 V
C.1.87 V
D.1.57 V
Solution
2Fe (s) + O2(g) + 4H+ (aq) → 2Fe+2 (aq) + 2H2O(l)
N = 4 (no. of moles of electron involved)
From Nernst′s equation,
Ecell = Eocell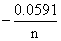 log Q
log Q
= 1.67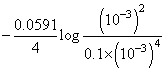 {∴ [H+] = 10-pH}
{∴ [H+] = 10-pH}
= 1.67 - 0.106 = 1.57 V
N = 4 (no. of moles of electron involved)
From Nernst′s equation,
Ecell = Eocell
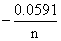 log Q
log Q= 1.67
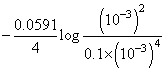 {∴ [H+] = 10-pH}
{∴ [H+] = 10-pH}= 1.67 - 0.106 = 1.57 V
Create a free account to view solution
View Solution FreeMore Aromatic Hydrocarbons Questions
Among P, Q, R and S, the aromatic compound(s) is / are :...Which of the following reactions is not shown by benzene -...Consider the following reaction :CH3CH2CH3 + Br →Which of the following types of hydrogen atoms are most easily ab...Debye force is present in: -...3-phenylpropene on reaction with HBr gives (as a major product) :-...