Aromatic HydrocarbonsHard
Question
Consider the following reaction :
CH3CH2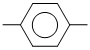 CH3 + Br →
CH3 + Br →
Which of the following types of hydrogen atoms are most easily abstracted by the bromine atom to form HBr ?
CH3CH2
 CH3 + Br →
CH3 + Br →Which of the following types of hydrogen atoms are most easily abstracted by the bromine atom to form HBr ?
Options
A.Hydrogen atoms attached to a benzene ring
B.Hydrogen atoms of the methyl group attached directly to a benzene ring
C.Hyrogen atoms of the methylene group of the ethyl substitutent
D.Hydrogen atoms of the methyl group of the ethyl substituent
More Aromatic Hydrocarbons Questions
An aromatic hydrocarbon (A) reacts with methyl chloride and anhy. AlCl3 to give an another hydrocarbon (B) whose vapour ...3-Chloro-4-methylbenzenesulphonic acid reacts with superheated steam to give-...Benzene on reaction with ′A′ forms which on reaction with ′B′ forms ′A′ and ′B...The final product of the reaction C6H6 + Cl2 is−..., Major product is -...