Carboxylic Acid and Acid DerivativesHard
Question
Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solution of density 1.15 g/mL.
The molarity of the solution is
The molarity of the solution is
Options
A.1.78 M
B.2.00 M
C.2.05 M
D.2.22 M
Solution
Total mass of solution = 1000 + 120 = 1120 g
Total volume of solution in (L)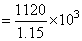
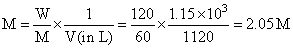
Total volume of solution in (L)
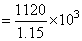
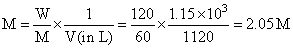
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
Acetic anhydride and ammonia gives the product :...Consider the following reaction: True about the above reaction is:...Which optically active compound on reduction with LiAlH4 will give optically inactive compound ?...CnH2n+1COCl represent the general formula of acyl chloride. Unstable member of this family would be obtained if we put -...The conductivity of 0.01 mol/dm3 aqueous acetic acid at 300K is 19.5 × 10-5 ohm-1 cm-1 and the maximum limiting mol...