Carboxylic Acid and Acid DerivativesHard
Question
The conductivity of 0.01 mol/dm3 aqueous acetic acid at 300K is 19.5 × 10-5 ohm-1 cm-1 and the maximum limiting molar conductivity of acetic acid at the same temperature is 390 ohm-1 cm2 mol-1. The degree of dissociation of acetic acid is.
Options
A.0.5
B.0.05
C.5 × 10-3
D.5 × 10-7
Solution
Molar conductance of 0.01 mol/dm3
∴ μ = k ×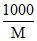 = 19.5 × 10-5 ×
= 19.5 × 10-5 × 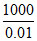
= 19.5 ohm-1 cm2 mol-1
∴ α =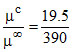 = 0.05
= 0.05
∴ μ = k ×
= 19.5 ohm-1 cm2 mol-1
∴ α =
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
-Ka will have the highest value for which of the following acids. -...Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solution of density 1.15 g/mL. The molarity of the solu...Amongst the acids,(i) CH ≡ CCOOH (ii) CH2 = CHCOOH and (iii) CH3CH2COOH, the acid strength follows the sequence...Which of the following reactions will give α-hydroxy acid as a product :...CH3COCl is used to detect the number of amino groups in the organic compounds. When a compound of molecular mass 180 is ...