Chemical BondingHard
Question
Some quantity of water is contained in a container as shown in figure. As neon is added to this system at constant pressure, amount of liquid water in the vessel -
(Assuming neon is not soluble in water)
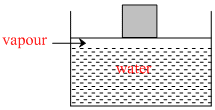
(Assuming neon is not soluble in water)

Options
A.Increases
B.Decreases
C.Remains same
D.Changes unpredictably