Chemical EquilibriumHard
Question
The equilibrium constant (KC) for the reaction N2(g) + O2(g) → 2NO(g) at temperature T is 4 × 10-4. The value of KC for the reaction. NO(g)→ 1/2 N2 (g) + 1/2 O2(g) at the sametemperature is :-
Options
A.50.0
B.0.02
C.2.5 × 102
D.4 × 10-4
Solution
N2 + O2 → 2NO KC = 4 × 10-4
2NO → N2 + O2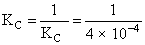
NO →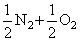
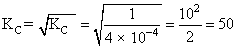
2NO → N2 + O2
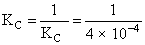
NO →
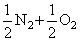
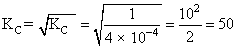
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the reaction 3 A (g) + B ⇋ (g) 2 C (g) at a given temperature, Kc = 9.0. What must be the volume of the flask,...For the following three reactions a, b and c, equilibrium constants are given: a. CO(g) + H2O(g) ⇋ CO2(g) + H2(g);...An aqueous solution of volume 500 ml, when the reaction 2Ag+ (aq) + Cu(s) $\rightleftharpoons$Cu2+ (aq) + 2Ag(s) reached...The equilibrium constant for the reaction N2(g) + O2(g)$\rightleftharpoons$ 2NO(g) is 4.0 × 10−4 at 2000 K. In the prese...The following equilibria are given : N2 + 3H2 ⇋ 2NH3 K1 N2 + O2 ⇋ 2NO K2 H2 + O2 ⇋ H2O K3 The equilibr...