ThermodynamicsHard
Question
The incorrect expression among the following is :-
Options
A.K = e-ᐃGo/RT
B.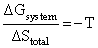
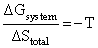
C.In isothermal process, Wrecersible = - nRT ln 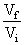
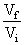
D.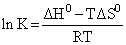
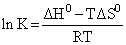
Solution
(1) Go = - RT ln K
K = e- ᐃGo/RT
(2)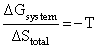
(3) In isothermal process Wrecersible = - nRT ln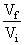
(4) ᐃGo = ᐃHo - TᐃS
- RTln K = ᐃHo - TᐃSo
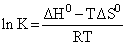
K = e- ᐃGo/RT
(2)
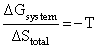
(3) In isothermal process Wrecersible = - nRT ln
(4) ᐃGo = ᐃHo - TᐃS
- RTln K = ᐃHo - TᐃSo
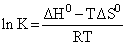
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The work involved (w) in an isothermal expansion of n moles of an ideal gas from an initial pressure of ‘P’ atm to final...Two moles of an ideal gas (Cv,m = 1.5R) is subjected to the following changes in states.$A\left( \text{500 K, 5 bar} \ri...The dissolution of NH4Cl in water is endothermic even through NH4Cl dissolves in water spontaneously. Which one of the f...For a process to occur under adiabatic conditions, the essential condition(s) is/are...For the cyclic process given below, which of the following relations are correct?...