Solid StateHard
Question
Lithium forms body centred cubic structure. The length of the side of its unit cell is 351 pm. Atomic radius of the lithium will be :-
Options
A.152 pm
B.75 pm
C.300 pm
D.240 pm
Solution
For BCC, r 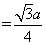 , a = 351 pm
, a = 351 pm
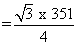 = 151.80 pm
= 151.80 pm
r = 151.80 pm
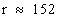 pm (approx)
pm (approx)
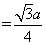 , a = 351 pm
, a = 351 pm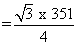 = 151.80 pm
= 151.80 pmr = 151.80 pm
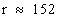 pm (approx)
pm (approx)Create a free account to view solution
View Solution FreeMore Solid State Questions
TiO2 is a well-known example of...A metallic element exists as cubic lattice. Each edge of the unit cell is 4.0 Å. The density of the metal is 6.25 g/cm3....Aluminium crystallizes in cubic system with unit cell edge length equal to 4.0 Å. If its density is $\frac{45}{16}$g/cm3...Which one of the following schemes of ordering closed packed sheets of equal sized spheres does not generate close packe...The radius of $\text{Ag}^+$ ion is $126\text{ pm}$ while that of $\text{I}^-$ ion is $216\text{ pm}$. The coordination n...