MetallurgyHard
Question
From the following statements regarding H2O2, choose the incorrect statement :
Options
A.It has to be stored in plastic or wax lined glass bottles in dark
B.It has to be kept away from dust
C.It can act only as an oxidizing agent
D.It decomposes on exposure to light
Solution
(1) H2O2 to be stored in plastic or wax lined glass in dark because, traces of alkali (present in glass) strongly catalyse the decomposition of H2O2 H2O2 → H2O + 1/2 O2
(2) H2O2 to be kept away from dust because dust induces explosive decomposition of H2O2.
(3) H2O2 can act as oxidising agent as well as reducing agent depending on condition, because the oxidation state of O-atom in H2O2 is −1. Which is in between highest (+2) and lowest (−2) oxidation state of O-atom.
(4) H2O2 decomposes in presence of light H2O2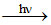 H2O + 1/2 O2
H2O + 1/2 O2
(2) H2O2 to be kept away from dust because dust induces explosive decomposition of H2O2.
(3) H2O2 can act as oxidising agent as well as reducing agent depending on condition, because the oxidation state of O-atom in H2O2 is −1. Which is in between highest (+2) and lowest (−2) oxidation state of O-atom.
(4) H2O2 decomposes in presence of light H2O2
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Magnesium is extracted by electrolysing fused magnesium chloride containing NaCl & CaCl2 using :...Which of the following ores is(are) concentrated by froth floatation?...Mac Arthur process is used for the extraction of :...In which of the following extration no reducing agent is required ?...In the cyanide extraction process of silver from argentite ore, the oxidising and reducing agents used are...