Carboxylic Acid and Acid DerivativesHard
Question
Reaction of 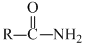 with a mixture of Br2 and KOH gives R - NH2 as the main product. The intermediates. involved in this reaction are :
with a mixture of Br2 and KOH gives R - NH2 as the main product. The intermediates. involved in this reaction are :
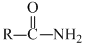 with a mixture of Br2 and KOH gives R - NH2 as the main product. The intermediates. involved in this reaction are :
with a mixture of Br2 and KOH gives R - NH2 as the main product. The intermediates. involved in this reaction are : Options
A.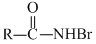
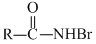
B.R - NHBr
C.R - N = C = O
D.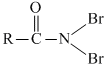
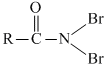
Solution
It is example of Hoffmann bromamide reaction.
RCONH2 + Br2 + 4KOH(aq.) → RNH2 + 2KBr + 2H2O + K2CO3
RCONH2 + Br2 → RCONHBr + HBr
RCONHBr + OH- →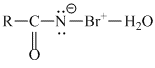
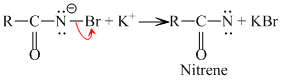

R - N = C = O + 2KOH → K2CO3
RCONH2 + Br2 + 4KOH(aq.) → RNH2 + 2KBr + 2H2O + K2CO3
RCONH2 + Br2 → RCONHBr + HBr
RCONHBr + OH- →
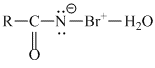
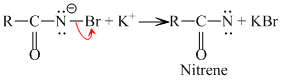

R - N = C = O + 2KOH → K2CO3
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
Why − OH group in ethyl alcohol is neutral, while it is acidic in acetic acid -...Acetic acid vapours when passed over aluminium phosphate forms -...Which one of the following esters cannot undergo self Claisen condensation?...Which of the following is the weakest acid ?...Cellulose upon acetylation with excess acetic anhydride/H2SO4 (catalytic) gives cellulose triacetate whose structure is...