ElectrochemistryHard
Question
The standard reduction potential data at 25oC is given below
Eo (Fe3+, Fe2+) = + 0.77 V ;
Eo (Fe2+, Fe) = - 0.44 V ;
Eo (Cu2+, Cu) = + 0.34 V ;
Eo (Cu+, Cu) = + 0.52 V ;
Eo (O2(g) + 4H+ + 4e- → 2H2O] = +1.23 V ;
Eo [(O2(g) + 2H2O + 4e- → 4OH- )] = +0.40 V ;
Eo (Cr3+, Cr) = -0.74 V ;
Eo (Cr2+, Cr) = -0.91 V ;
Match Eo of the redox pair in List-I with the values given in List-II and select the correct answer using the code given below the lists :
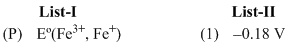
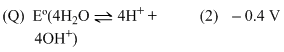
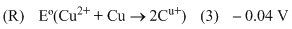

Eo (Fe3+, Fe2+) = + 0.77 V ;
Eo (Fe2+, Fe) = - 0.44 V ;
Eo (Cu2+, Cu) = + 0.34 V ;
Eo (Cu+, Cu) = + 0.52 V ;
Eo (O2(g) + 4H+ + 4e- → 2H2O] = +1.23 V ;
Eo [(O2(g) + 2H2O + 4e- → 4OH- )] = +0.40 V ;
Eo (Cr3+, Cr) = -0.74 V ;
Eo (Cr2+, Cr) = -0.91 V ;
Match Eo of the redox pair in List-I with the values given in List-II and select the correct answer using the code given below the lists :
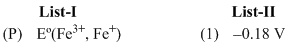
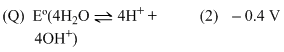
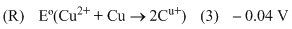

Options
A.P → 4, Q → 1, R → 2, S → 3
B.P → 2, Q → 3, R → 4, S → 1
C.P → 1, Q → 2, R → 3, S → 4
D.P → 3, Q → 4, R → 1, S → 2
Solution
Detailed solution available for registered users.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
A solution containing 1.0 M each of Cu(NO3)2, Mg(NO3)2, AgNO3, Hg(NO3)2 is being electrolysed using inert electrodes. Th...In which of the following cell (s) : Ecell = Eocell ?...If the cell reaction is spontaneous then...A dilute aqueous solution of Na2SO4 is electrolysed using platinum electrodes. The products at the anode and cathode are...The EMF of the cell: Zn|Zn2+ (0.01 M)||Fe2+ (0.001 M)|Fe at 298 K is 0.2905 V, then the value of equilibrium constant fo...