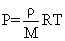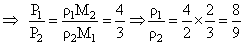JEE Advanced | 2013KTGHard
Question
Two non-reactive monoatomic ideal gases have their atomic masses in the ratio 2 : 3. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is 4 : 3. The ratio of their densities is :-
Options
A.1 : 4
B.1 : 2
C.6 : 9
D.8 : 9
More KTG Questions
The average translational kinetic energy of molecule depends on :-...Following is not a postulate of kinetic theory of gases :-...One mole of a gas is subjected to two process AB and BC, one after the other as shown in the figure. BC is represented b...A cylinder of 200 litre capacity is containing H2. The total average translational kinetic energy of molecules is 1.52 &...A box contains N molecules of a gas. If the number of molecules is doubled, then the pressure will:-...