JEE Advanced | 2013General Organic ChemistryHard
Question
The hyperconjugative stbilities of tert-butyl cation and 2-butene respectively, are due to
Options
A.
σ → P (empty) and σ → π* electron delocalisations
B.σ → σ* and σ → π electron delocalisations
C.σ → p (filled) and σ → π electron delocalisations
D.
p (filled) → σ* and σ → π* electron delocalisations
Solution
Hyperconjugative stabilities of ter-butyl cation [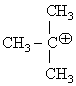 ] and 2-Butene [CH3-CH=CH-CH3]
] and 2-Butene [CH3-CH=CH-CH3]
involves 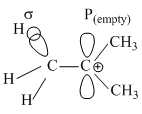 and
and 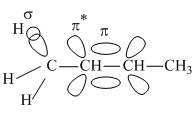 respectively
respectively
π*=Antibonding molecular orbital
π=Bonding molecular orbital
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
In the following compounds, anisole (I), benzene (II) and nitrobenzene (III), the ease of reaction with electrophiles is...Due to the presence of an unpaired electron, free radicals are:...Which is most reactive for SN1 reaction :-...Which one is most reactive towards SN1 reaction ?...Which one of the following compounds is resistant to nucleophic attack by hydroxylions...