JEE Advanced | 2013Coordination CompoundHard
Question
The pair(s) of coordination complex/ion exhibiting the same kind of isomerism is(are) -
Options
A.[Cr(NH3)5Cl]Cl2 and [Cr(NH3)4Cl2]Cl
B.[Co(NH3)4Cl2]+ and [Pt(NH3)2(H2O)Cl]+
C.[CoBr2Cl2]2- and [PtBr2Cl2]2-
D.[Pt(NH3)3(NO3)] Cl and [Pt(NH3)3Cl] Br
Solution
(A) [Cr(NH3)5Cl]Cl2 does not show any isomerism while [Cr(NH3)4Cl2]Cl shows geometrical isomerism
(B) [Co(NH3)4Cl2]+ and [Pt(NH3)2(H2O) (Cl)]+
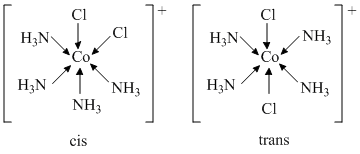
hence, [Co(NH3)4Cl2]+ ion shows geometrical isomerism.
[Pt(NH3)2(H2O) (Cl)]+
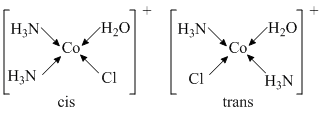
hence, [Pt(NH3)2(H2O)(Cl)]+ ion shows geometrical isomerism
(C) [CoBr2Cl2]2- and [PtBr2Cl2]2-
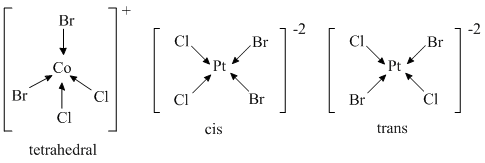
does not show any isomerism square planar and shows geometrical isomerism
(D) [Pt(NH3)3(NO3)]Cl and [Pt(NH3)3Cl]Br
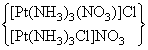 show ionisation isomerism
show ionisation isomerism
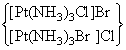 show ionisation isomerism
show ionisation isomerism
hence, Ans is (B, D)
(B) [Co(NH3)4Cl2]+ and [Pt(NH3)2(H2O) (Cl)]+

hence, [Co(NH3)4Cl2]+ ion shows geometrical isomerism.
[Pt(NH3)2(H2O) (Cl)]+
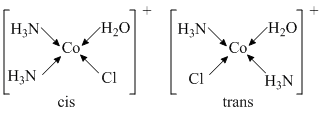
hence, [Pt(NH3)2(H2O)(Cl)]+ ion shows geometrical isomerism
(C) [CoBr2Cl2]2- and [PtBr2Cl2]2-

does not show any isomerism square planar and shows geometrical isomerism
(D) [Pt(NH3)3(NO3)]Cl and [Pt(NH3)3Cl]Br
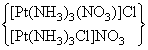 show ionisation isomerism
show ionisation isomerism 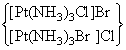 show ionisation isomerism
show ionisation isomerismhence, Ans is (B, D)
Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
White silver surface turns black when O3 is passed over it. This is due to the formation of...Given below are two statements :Statement I: $\left\lbrack {CoBr}_{4} \right\rbrack^{2 -}$ ion will absorb light of lowe...Which of the following pairs of name and formula of complexes, is correct ?...The disodium salt of ethylenediaminetetracetic acid can be used to estimate the following ion(s) in the aqueous solution...The correct option(s) regarding the complex [Co(en) (NH3)3(H2O)]3+ :- (en = H2NCH2CH2NH2) is /are...